Nach US-Präsident Trump scheint auch Präsident Macron an Hydroxychloroquin interessiert
US-Präsident Donald Trump könnte Recht behalten mit seiner Notfallzulassung des deutschen Arzneimittels Resochin. Nach US-Präsident Trump scheint auch Präsident Macron an Hydroxychloroquin interessiert. Ärzte in Frankreich und Amerika haben Erfolge in der Behandlung von Covid-19 mit dem Medikament signalisiert. Bayer hatte den USA drei Millionen Tabletten des Malaria-Medikaments mit dem Wirkstoff Chloroquin gespendet. Deutschland hat sich das Medikament ebenfalls gesichert.

After US President Trump, President Macron also seems interested in hydroxychloroquine
[UPDATE: According to a Chinese publication dated February 19, 2020, a COVID-19 study with chloroquine in ten Chinese hospitals, more than 100 COVID-19 patients has produced encouraging results. Chloroquine is a low-cost malaria drug that has been used for about 80 years.] In a further study conducted in China and France, two dozen coronavirus patients were also found to be very well cured with chloroquine. In China and France, two dozen coronavirus patients have already been treated with chloroquine with very good results. China has already included chloroquine in its recommendations for the prevention and treatment of COVID-19 pneumonia. UPDATE: Belgium has also included chloroquine/hydroxychloroquine in its treatment recommendations for COVID-19. The same applies to the treatment guideline for Covid-19 in South Korea]. US President Donald Trump sees this as a major breakthrough in the fight against the coronavirus. Against strong resistance, the U.S. President had approved Bayer's drug in the United States with an emergency marketing authorization. [UPDATE: At a press conference on 5 April 2020, US President Donald Trump reiterated his optimism about the potential of the antimalarial drug to treat COVID-19 patients and that the US government has stockpiled around 29 million tablets. In Switzerland, a fast-track approval for treatment with the active ingredient hydroxychloroquine is also under review. As german manager magazin reports on April 2, 2020, Bayer AG now intends to produce chloroquine in Europe.]
[Update: On March 31, 2020, China submitted a further study with 62 COVID-19 patients to Wuhan University showing faster recovery with hydroxychloroquine (HCQ)].
As the Wall Street Journal wrote on Sunday, doctors in Kansas City reported successes in Covid-19 patients treated with the active ingredient chloroquine. The demand for the drug has already risen sharply in the USA. [On 20 March 2020, the Swedish pharmaceutical company Recipharm reported a similar increase in demand for the product Klorokinfosfat RPH Pharma (chloroquine phosphate) which they market in Sweden. The company cited as a reason the potential use of chloroquine in the treatment of patients with coronavirus-COVID-19-induced pneumonia].
One of the leading French virologists, Professor Didier Raoult, M.D., head of the Department of Infectious Diseases at La Timone Hospital in Marseille, France, also spoke of a clear effect in a second study. This is reported in the French newspaper Le Monde. The new study includes 80 patients, 65 (81 percent) of whom, according to the authors, show a "favourable development" and were discharged from hospital after less than five days on average. The French government has subsequently officially approved the drug chloroquine for treatment in COVID-19 patients.
The patients were treated with Covod-19 with
- Hydroxychloroquine (200 mg x 3 per day for 10 days)
- and azithromycin (500 mg on day 1, then 250 mg per day for another 5 days).
A broad-spectrum antibiotic is also used for severe pneumonia.
UPDATE France:
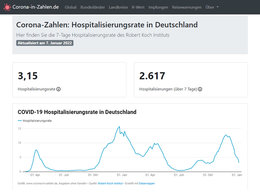
In the night of March 30, 2020, the IHU - Méditerranée Infection in Marseille reported only one death COVID-19 patient among 1,291 coronavirus patients (Update 13.05.2020: 3.287 patients, 17 died). treated with hydroxychloroquine and azithromycin. All COVID-19 patients in the chloroquine-azithromycin group have been treated with the two drugs for at least 3 days. In the other hospitals in Marseille, however, 16 of 2,222 COVID-19 patients died (Update 13.05.2020: 4844 patients, 151 died) without treatment with chloroquine and azithromycin.
According to TheGuardian and Bloomberg following US President Donald Trump, French President Emmanuel Macron now also seems to be interested in the malaria drug hydroxychloroquine. The French president said after a visit to the French virologist Professor Dr. Didier Raoult that he would support Didier Raoult's treatment and have it tested. The infection specialist Professor Didier seems to be very successful in treating more than 3000 corona patients with a combination of the malaria drug Hydroxychloroquine and the antibiotic Azithromycin.
The German chemical and pharmaceutical group Bayer AG has donated three million tablets of the malaria drug with the active ingredient chloroquine to the USA. French media report that the French pharmaceutical company Sanofi also intends to make the drug for treatment against COVID-19 available to the French authorities in sufficient quantities. The swiss Novartis AG donates up to 130 million doses of generic hydroxychloroquine to support the global COVID-19 pandemic response. UPDATE: In the USA another Big Pharma company donated 400,000 tablets of the drug hydroxychloroquine on March 31, 2020. The American pharmaceutical giant Amneal Pharmaceuticals, Inc. has donated the malaria drug to the US state of Louisiana for the treatment of coronaviruses. Teva Pharmaceutical Industries already announced a donation of more than 6 million doses of hydroxychloroquine sulfate tablets to hospitals in the US less than two weeks ago to meet the urgent demand for the drug as an investigational target for the treatment of COVID-19. Within one month, the American-Israeli pharmaceutical company also plans to make available another 10 million doses in the USA].
UPDATE USA:
According to a report in the American newspaper THEHILL on 20 April 2020, the US Food and Drug Administration (FDA) and Novartis have reached an agreement enabling the Swiss pharmaceutical company to conduct a clinical trial with hydroxychloroquine for patients with COVID-19, the disease caused by the novel coronavirus. As reported by the German TV channel ARD, the American news channel CNN an the british newspaper The Guardianon on 19.05.2020, US President Trump says he is taking the malaria drug hydroxychloroquine as a preventive measure against Covid-19 because of the corona pandemic. Trump gives as a reason that many doctors and nurses would also take it as a precaution. He also takes zinc to strengthen his immune system. [Editor's note: The malaria drug hydroxychloroquine seems to act as a zinc ionophore and increase intracellular zinc concentrations. Zinc in turn has an inhibitory effect on the RNA polymerase of coronaviruses].
UPDATE Canada:
The Canadian associated company Codebase Ventures Inc. has secured the rights to the Transdermal Hydroxychloroquine Drug Delivery System, as it announced in a press release dated April 20, 2020.
UPDATE India:
India produces 70 percent of the world's supply of hydroxychloroquine. As the newspaper theeconomictimes reported on 16 April 2020, India is already sending hydroxychloroquine to 55 countries affected by coronavirus. India supplies the drug Hydroxychloroquine to the neighbouring countries Afghanistan, Bhutan, Bangladesh, Nepal, Maldives, Mauritius, Sri Lanka and Myanmar. Further deliveries go to Zambia, Dominican Republic, Madagascar, Uganda, Burkina Faso, Niger, Mali Congo, Egypt, Armenia, Kazakhstan, Equador, Jamaica, Syria, Ukraine, Chad, Zimbabwe, France, Seychelles, Jordan, Kenya, Netherlands, Nigeria, Oman and Peru. Hydroxychloroquine is also shipped to the Philippines, Russia, Slovenia, South Africa, Sri Lanka, Tanzania, United Arab Emirates, Uzbekistan, Urugway, Colombia, Algeria, Bahamas, Mauritius and the United Kingdom. India had temporarily banned exports because the USA ordered 29 million doses and Hydroxychloroquine is partly used preventively by medical staff in India.
UPDATE Australia:
Australian researchers start a clinical trial of COVID-19 in 50 hospitals in Australia. The COVID-19 patients will be treated with the malaria drug chloroquine and the HIV drug Kaletra (Lopinavir / Ritonavir). "We are now ready to start patient trials with the drugs, one of which is an HIV drug and the other an anti-malaria drug," said Professor David Paterson of the University of Queensland. He is director of the University of Queensland's Centre for Clinical Research and a consultant on infectious diseases at the Royal Brisbane and Women's Hospital (RBWH). "Prior to the start of clinical trials, the drugs were administered to some of the first COVID-19-infected patients in Australia, and all have made a full recovery without leaving any traces of the virus in their systems". The Australian Minister of Health Greg Hunt promised on 2 April 2020 that the COVID-19 drug hydroxychloroquine will soon be available in Australia in any hospital that wishes to use it.
COVID-19-Interview on 7NEWS Australia
Professor David Paterson, 16.03.2020
As reported by the newspaper Welt, Germany has also secured "larger quantities" of the potential Covid-19 drug, according to German Health Minister Jens Spahn (CDU). [UPDATE: According to a report of the SWR of March 18, 2020, the drug has already been used in China and Italy for weeks in Covid-19 patients. In Germany, despite 80 years of experience with the drug, it is not approved for the treatment of patients with coronavirus. As the newspaper BILD reported on 3 April 2020, Jens Spahn is also pinning his hopes on the malaria drug Resochin from the pharmaceutical company Bayer to combat medically severe corona disease.
According to the news channel Phoenix, Wolf-Dieter Ludwig, Chairman of the German Medical Commission, also considers two active ingredients to be promising: The first is Remdesivir, which was developed as a remedy for Ebola and is now to be tested in two large studies on corona patients in Germany. The second is chloroquine, or hydroxychloroquine, "a very well known malaria drug that probably also has antiviral activity and is therefore able to inhibit the activity of SARS CoV-2 viruses," said Ludwig. According to Ludwig, these two active substances are in first and second place in the assessment of the commission to the Federal Ministry of Health].
COVID-19-Interview on german SWR
Prof. Peter Kremsner, 18.03.2020
Download [PDF, 3 Pages - 215 KB]
SARS-CoV-2 Covid-19 Studie 2020
Chloroquine and hydroxychloroquine as available weapons to fight COVID-19
More information about Chloroquine and Hydroxychloroquine
Chloroquine, a widely-used anti-malarial and autoimmune disease drug, has recently been reported as a potential broad-spectrum antiviral drug. Chloroquine is known to block virus infection by increasing endosomal pH required for virus/cell fusion, as well as interfering with the glycosylation of cellular receptors of SARS-CoV. Besides its antiviral activity, chloroquine has an immune-modulating activity, which may synergistically enhance its antiviral effect in vivo. Chloroquine is a cheap and a safe drug that has been used for more than 70 years and, therefore, it is potentially clinically applicable against the 2019-nCoV. Hydroxychloroquine acts like chloroquine, but is less harmful to the retina than chloroquine. Both agents increase the supply of zinc, which is important for the immune system.